

Slide Background
iTraceNet
QRCode label generating and printing solution is designed to meet the Central Drugs Standard Control Organization (CDSCO) regulation and ease business operations ensuring to comply with the regulation compliances.
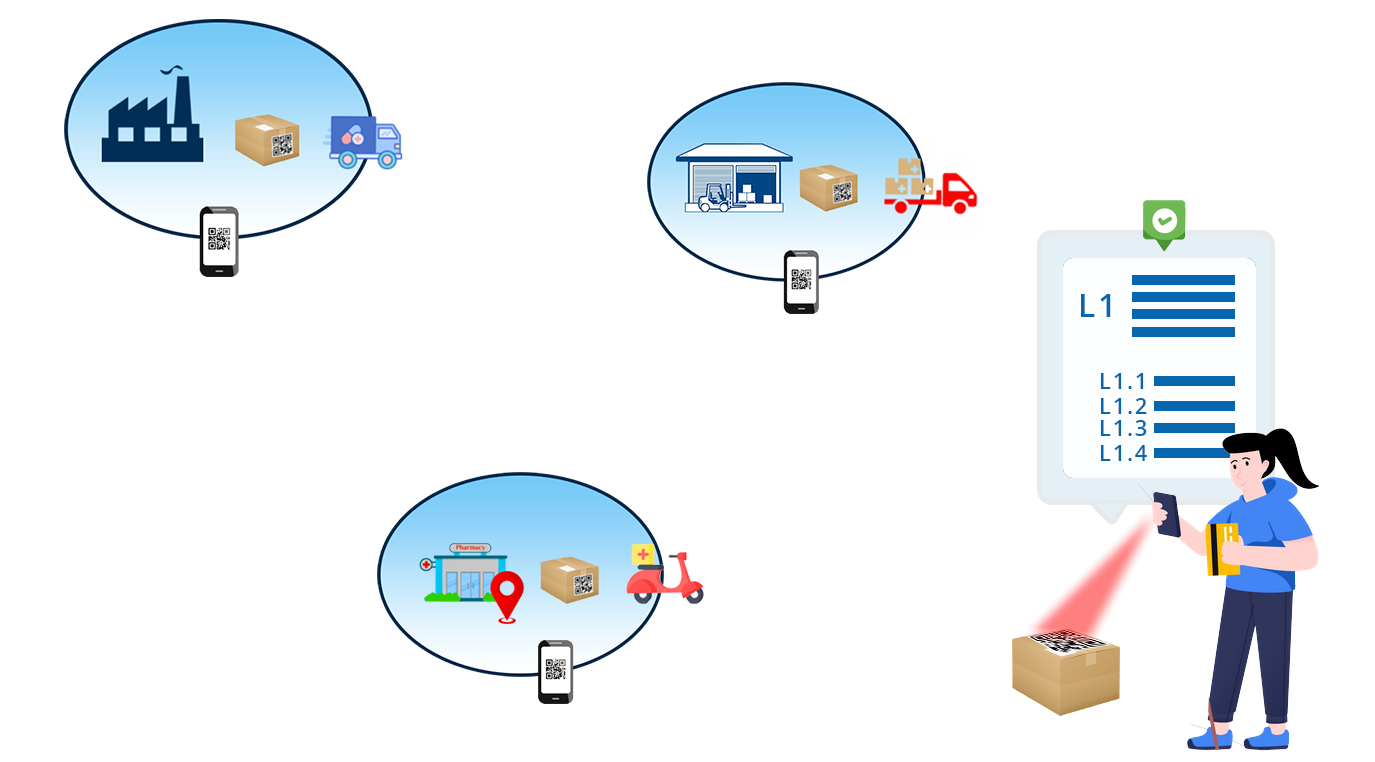
Build & Design
Guidelines
As per Govt. Gazette CG-DL-E-19012022-232768 enabling QR code based traceability of API materials is mandatory from 1st Jan 2023. In the Drugs Rules, 1945, in rule 96, after sub-rule (4), following sub-rule shall be inserted, namely:— “(5) Every active pharmaceutical ingredient (bulk drug) manufactured or imported in India shall bear Quick Response code on its label at each level packaging that store data or information readable with software application to facilitate tracking and tracing.

iTraceNet
You are with complete information about the product you purchase/consume with authenticity. It allows the supply chain networks, and the companies and people collaborating across them, to meet unique business needs and solve critical supply chain challenges

Add Your Heading Text Here

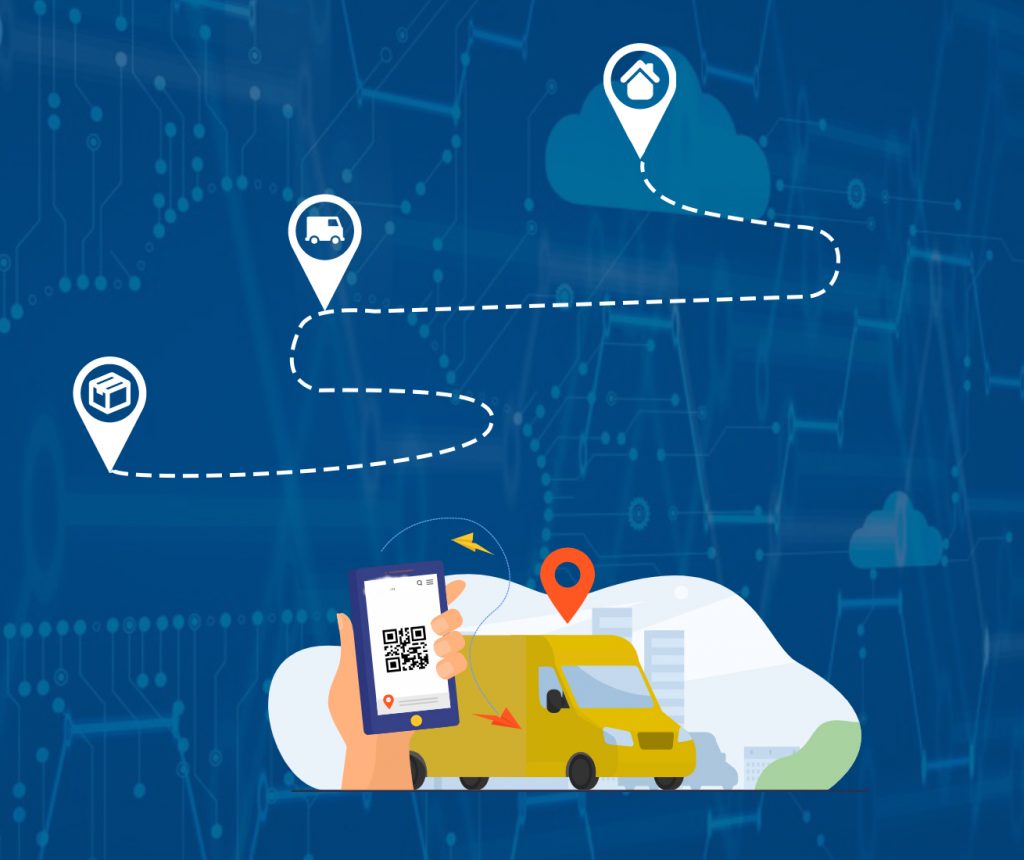
Issue customized risk assessments about packages

Track package
Package of complete integrity

GS1 Standards for QR Generating
Analytics Capabilities

